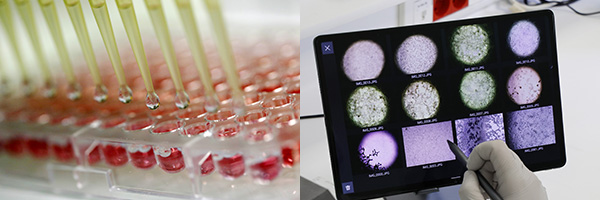
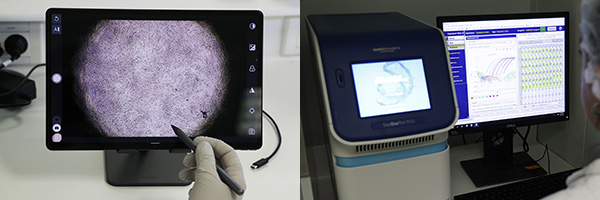
Department: Testing Services for management of viral contamination

We develop experimental strategies and carry out virucidal activities tests considering the real use conditions of disinfectant products and equipment in order to optimize the management of the viral contamination for breeding industries, research laboratories, R&D centers, Biotech and industrial production areas.
- Experimental procedures consider:
- Management process of microbiological disinfection and client's products/equipment
- Dirty conditions associated with chemical and organic pollutants of site-specific activities and environment
- Viral strain of client or a wide range of human and animal viral strains.
- Standardized experimental procedures if applicable